Understanding atomic radius is one of the most important steps in mastering basic chemistry. Whether you’re a student preparing for exams or just trying to make sense of the periodic table, this concept helps you understand how atoms behave, bond, and react.
In this complete, easy-to-read guide, you’ll learn:
- What atomic radius is (simple definition)
- It’s different Types
- Periodic trends across the periodic table
- Key factors affecting atomic size
- Real-world examples and comparisons
What is Atomic Radius?
Definition (Simple)
It is the distance from the center of an atom’s nucleus to the outermost boundary of its electron cloud.
In simpler terms:
It tells you how big an atom is.
However, atoms don’t have a sharp boundary like a solid sphere, so scientists define it’s radius in practical ways (which we’ll cover in types).
Why it is Important?
It helps us understand:
- How atoms bond with each other
- The reactivity of elements
- Trends in the periodic table
- Differences between metals and non-metals
For example:
- Larger atoms tend to lose electrons easily
- Smaller atoms tend to attract electrons strongly
Types of Atomic Radius
Since atoms don’t have a fixed edge, scientists use different methods to measure atomic size.
1. Covalent Radius
The covalent radius is half the distance between the nuclei of two identical atoms bonded together.
📌 Example:
In a hydrogen molecule (H₂), the distance between two hydrogen atoms is measured, and half of it is the covalent radius.
2. Ionic Radius
The ionic radius refers to the size of an atom when it has gained or lost electrons (forming an ion).
- Cations (positive ions) → Smaller radius
- Anions (negative ions) → Larger radius
📌 Example:
- Na⁺ is smaller than Na
- Cl⁻ is larger than Cl
3. Metallic Radius
The metallic radius is half the distance between the nuclei of two adjacent metal atoms in a metallic structure.
📌 Common in metals like iron, copper, etc.
4. Van der Waals Radius
The van der Waals radius is half the distance between two non-bonded atoms.
📌 Used when atoms are close but not chemically bonded.
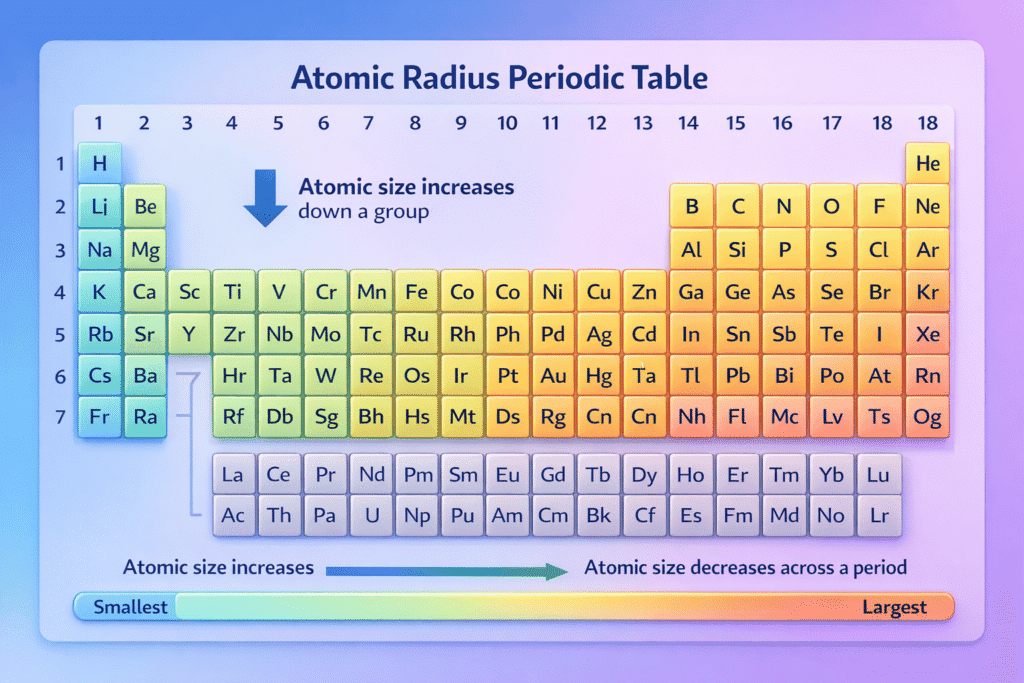
Atomic Radius Trends in the Periodic Table
Understanding trends makes everything easier. Let’s break it down clearly:
Trend 1: It Increases Down a Group
As you move down a group (column):
- Atomic size increases
Why?
- More electron shells are added
- Electrons are farther from the nucleus
- Shielding effect increases
📌 Example:
- Lithium < Sodium < Potassium
Trend 2: It Decreases Across a Period
As you move left to right across a period (row):
- Atomic size decreases
Why?
- More protons are added to the nucleus
- Stronger nuclear attraction pulls electrons closer
- No new shell is added
📌 Example:
- Carbon > Nitrogen > Oxygen > Fluorine
Quick Memory Trick
- ⬇️ Down a group → Bigger atoms
- ➡️ Across a period → Smaller atoms
Factors Affecting Atomic Radius
Several factors determine the size of an atom:
1. Number of Electron Shells
More shells = larger atom
2. Nuclear Charge
Higher number of protons = stronger pull on electrons = smaller atom
3. Shielding Effect
Inner electrons block the pull of the nucleus on outer electrons.
More shielding = larger radius
4. Electron-Electron Repulsion
Electrons repel each other, pushing the outer electrons further away.
Atomic Radius vs Ionic Radius
| Feature | Atomic Radius | Ionic Radius |
|---|---|---|
| Definition | Size of neutral atom | Size of ion |
| Electron count | Balanced | Gained or lost |
| Size change | Fixed trend | Can increase/decrease |
📌 Key Insight:
- Cations are smaller
- Anions are larger
Real-Life Applications
1. Chemical Bonding
Helps predict bond length and strength
2. Reactivity
- Larger atoms → more reactive metals
- Smaller atoms → more reactive non-metals
3. Material Science
Determines structure of metals and crystals
4. Pharmaceuticals
Affects how atoms interact in drug molecules
Example-Based Understanding
Let’s simplify with a quick comparison:
Example 1: Sodium vs Chlorine
- Sodium → larger radius
- Chlorine → smaller radius
So sodium loses electrons easily, chlorine gains electrons.
Example 2: Magnesium vs Calcium
- Calcium is below magnesium
So calcium has a larger atomic radius
Common Mistakes Students Make
- Thinking atomic radius always increases → ❌
- Ignoring nuclear charge → ❌
- Confusing atomic and ionic radius → ❌
Summary (Quick Revision)
- Atomic radius = size of an atom
- Measured in different ways (covalent, ionic, metallic)
- Increases ↓ down a group
- Decreases → across a period
- Affected by nuclear charge, shielding, and shells
FAQs (People Also Ask)
It is the size of an atom measured from the nucleus to the outermost electron.
Because the number of protons increases, pulling electrons closer to the nucleus.
Elements at the bottom-left of the periodic table (like cesium).
Atomic radius is for neutral atoms, while ionic radius is for charged atoms (ions).